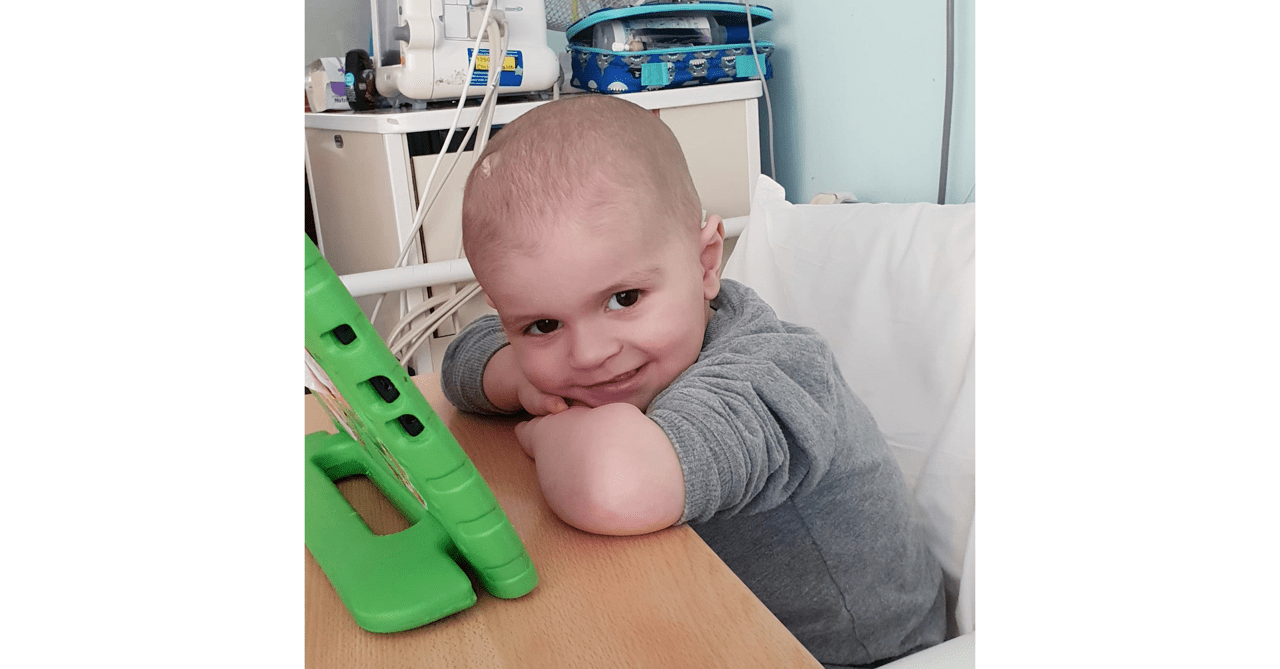
As with many other charities, organisations, supporter groups etc., 2020 certainly knocked us at Make William Well off our stride as those of you who follow us may have noticed by the lack of blog posts over the last year. In 2021 though we’re hoping to get back on track and if all goes to… Continue reading We’re Back…
Blog
CBD Project – Further Developments
Taken from a report produced by The University of Nottingham – original can be downloaded here Is there a role for Cannabidiol in the treatment of Children’s Brain Tumours? Project Update November 2019 Make William Well has generously supported a research project at the Children’s Brain Tumour Research Centre examining “Is there a role for… Continue reading CBD Project – Further Developments
UK Paediatric Brain Tumour Symposium – REVIEW
Introduction So, a week last Wednesday the 2019 UK Paediatric Brain Tumour Symposium took place in Nottingham. The venue was different to last year and unfortunately (well, fortunately for camera-shy yours truly) it was not recorded this time It was great meeting other parents (one of whom came all the way from America) as well… Continue reading UK Paediatric Brain Tumour Symposium – REVIEW
Brain Tumour Symposium
Less than a week to go until the second ever UK Paediatric Brain Tumour Symposium and I’m still struggling to comprehend the fact that I’ll be sharing a stage with the likes of Prof’s Grundy and O’Sullivan, both of whom I have the utmost respect for Although I have spoken previously about our journey, I… Continue reading Brain Tumour Symposium
UK Paediatric Brian Tumour Symposium 2019
I probably wouldn’t have been up for this had William’s scan not been stable but I’m happy to say I’ll be presenting at this year’s UK Paediatric Brain Tumour Symposium in Nottingham I attended the first ever one held last year and it was really informative. This year topics include cannabinoids, the ketogenic diet and… Continue reading UK Paediatric Brian Tumour Symposium 2019
Different Cannabinoids and Different Cancers
We have suspected for a while that different cannabinoids (CBD, THC etc.) have different effects on different types of cancer and this article supports that theory (it has been added it to the list of Academic Research Papers that we have been keeping on Cannabinoids, the Ketogenic Diet and Cancer) We were over the moon… Continue reading Different Cannabinoids and Different Cancers
Generous Donation in Memory of Tommy
As well as to raise awareness and money for more research into childhood brain tumours, we set up Make William Well to provide a resource for other families going through this journey As such, we receive regular requests for help and we do all we are able to. One of the hardest things about this, on many… Continue reading Generous Donation in Memory of Tommy
1000 Likes!
A massive Thank You to all those who have liked our Facebook Page and stuck with it! Every Like makes us feel more and more like what we’re doing is worthwhile and gives us the confidence to continue sharing our story and trying to help William and other kids benefit from what he has been… Continue reading 1000 Likes!
A Cure Can’t Wait
Ok, so donations are starting to come in at our JustGiving Team Page (thank you so much to everyone who has donated so far) and in just over two weeks time we’ll be setting off on our Hadrian’s Wall Challenge (fingers crossed for better weather than today!) This year we’ve teamed up with The Brain… Continue reading A Cure Can’t Wait
All Party Parliamentary Group on Brain Tumours – the Ketogenic Diet
I attended this event last week – it was held at Portcullis House just opposite Big Ben and the Houses of Parliament. I was invited to The All Party Parliamentary Group on Brain Tumours, basically a meeting held to discuss brain tumours attended by various members of parliament, by the charity Brain Tumour Research. During… Continue reading All Party Parliamentary Group on Brain Tumours – the Ketogenic Diet