The text and images below are taken from a report issued by the Children’s Brain Tumour Research Centre to those supporters of the CBD Research Project we proposed and helped fund…
 In vitro evaluation of the effect of cannabidiol as an adjuvant therapy for paediatric brain tumours: Update December 2018
In vitro evaluation of the effect of cannabidiol as an adjuvant therapy for paediatric brain tumours: Update December 2018
Over the last six months, work on the project has progressed at a pace. Most of the cell lines under investigation have been growing very well and therefore substantial progress has been made. A medical student join our team between September – December 2018, which increases our research capacity and also helps us raise awareness of brain tumours and cannabidiol research amongst clinicians of the future.
The experiments are being conducted on cells with a lineage from paediatric high grade glioma or paediatric ependymoma. These are the most robust cells in culture and have a faster growth rate, enabling more experiments to be performed on them per week.
For each experiment to be valid we have to perform it a minimum of 3 times (optimisation experiments) and 6 times for experimental data. Importantly, we have been able to show that we can kill tumour cells with CBD whilst the astrocyte population is unchanged.
Professor Richard Grundy, is pleased with the results, but adds a note of caution, “these results, though promising are very preliminary and there are a range of increasingly complex studies that need to be conducted to assess whether cannabinoids may have a role in treating children’s brain tumours”.
We are conducting a range of experiments to explore what happens to tumour cells when they are treated with CBD, and if the cells die, what is it that causes this to happen. Below is a summary of all of the different experiments being performed:
1. Metabolism Analysis
Metabolism assays are used to quantitatively measure the effect of a drug on the proliferation of cells i.e. how quickly the cells are growing and dividing. We have conducted a range of experiments so that we can extrapolate the concentration of the drug needed to achieve 50% cell death. The concentration of drug which causes 50% cell death (EC50) has been confirmed in the SF188, BxD-1425EPN and astrocytoma cells and this is then used for further experiments.
2. Western Blot
Western blots are used to detect specific proteins within cells. In our experiments, the cells were cultured either alone, in the vehicle the drug was diluted in, or in CBD. The cells were also cultured in the presence of two different control drugs, staurosporine (initiates apoptosis) or chloroquine (initiates autophagy) as controls. We then monitored the cells, and used Western blot analysis to identify which proteins are released during cell death. By understanding the clear mode of cell death, we are able to begin understanding how the different drugs cause the cells, or parts of the cell, to die.
3. 3D Spheroids
3D spheroid culture is thought to better represent the growth of brain tumours in vivo. In the CBTRC we have developed a model for culturing cells in 3D which allows end point evaluation by immunohistochemistry (IHC) a technique usually reserved for tissue pieces. So far, we have just grown the cells which have lineage as high grade glioma into 3D models as these cells rapidly divide and therefore can be used in many experiments at once. 3D evaluation has shown a significant difference between control cells grown in culture media only and those grown in CBD. We have prepared the spheroids in a way which means we can perform IHC at a later date to look for the markers of cell death (apoptosis and autophagy) and cell replication and also to identify their specific locations within the 3D structure.

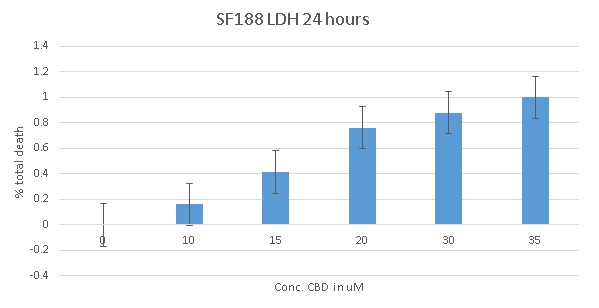
4. Lactate Dehydrogenase Assay
Lactate dehydrogenase (LDH) is a soluble enzyme present in most cells which is released into the culture medium upon cell death due to damage of the plasma membrane. The breakdown of the membrane may be due to either necrosis, or in smaller amounts, apoptosis or autophagy. To date we have completed this assay on the SF188 cells, with the BXD-1425EPN cells expected to be completed by Christmas. Figure 2 shows the increase in cell death which occurs with increasing concentrations of CBD over a 24 hour period.
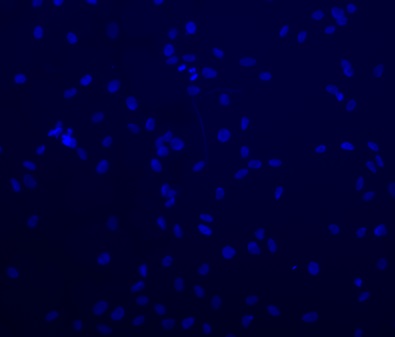
5. Immunofluorescence
Immunofluorescence is the detection of proteins using fluorescent markers. We aim to use the same antibodies that we have used in the Western blots to demonstrate by a second method the mode of cell death. Figure 4 shows a representative image of SF188 cells grown for 24 hours with the nuclei of the cells stained by DAPI to enable optimisation of cell numbers. This method ensures enough cells had been seeded to still be visible after any experimentation.
6. RNA Analysis
The two different groups of cells have been grown in the different concentrations of CBD, and a vehicle only solution. The cells have been prepared for RNA analysis. Gene expression analysis will be performed early 2019 to see if growing the cells in CBD alters the expression of different genes.
7. Hypoxic Chamber
It was an aim of the project to perform all of the experiments in normoxic conditions and in hypoxic conditions. Cell culture is usually performed at a constant temperature (37°C), high humidity and 5% carbon dioxide for the optimum growth of the cells. However, this is thought not to be a very good comparison to what is occurring in a brain tumour where the cells, particularly towards the centre of the tumour, are undergoing a degree of hypoxia or lack of oxygen. We have recently been granted access to a hypoxic chamber which allows us to run experiments at 1% oxygen which more closely represents the oxygen levels within the core of a tumour. To use this chamber cells have to be grown in antibiotic media and to demonstrate that they are mycoplasma free. We are still experiencing some infection difficulties with the diffuse infantile pontine gliomas (DIPG) cell lines, which is preventing us using the hypoxic chamber at present with those lines. We hope that by Christmas we will have these established, free from infection and experiments to be started on them in the New Year. We have now begun to explore the concentration of drug which causes 50% cell death on these cells in hypoxic conditions.
8. Future Plans
This year, preliminary experimental results were presented at BNOS 2018 and ISPNO 2018 in poster format. The first half of 2019 will be spent conducting further experiments, and we intend to share our results internationally via publication and at international conferences in 2019 and 2020.
We are discussing our preliminary results with colleagues at the University of Western Australia. They are also conducting cannabinoid research on paediatric brain tumours, but using cells from Medulloblastoma tumours. This is a great opportunity to compare results, and also gain an understanding from a wider range of tumours, and has the potential to be an exciting collaboration as the study progresses.
On behalf of the research team, we would like to thank you and your supporters for making this study possible. We are excited about the results and the potential impact we can share with the scientific community later in 2019.
